 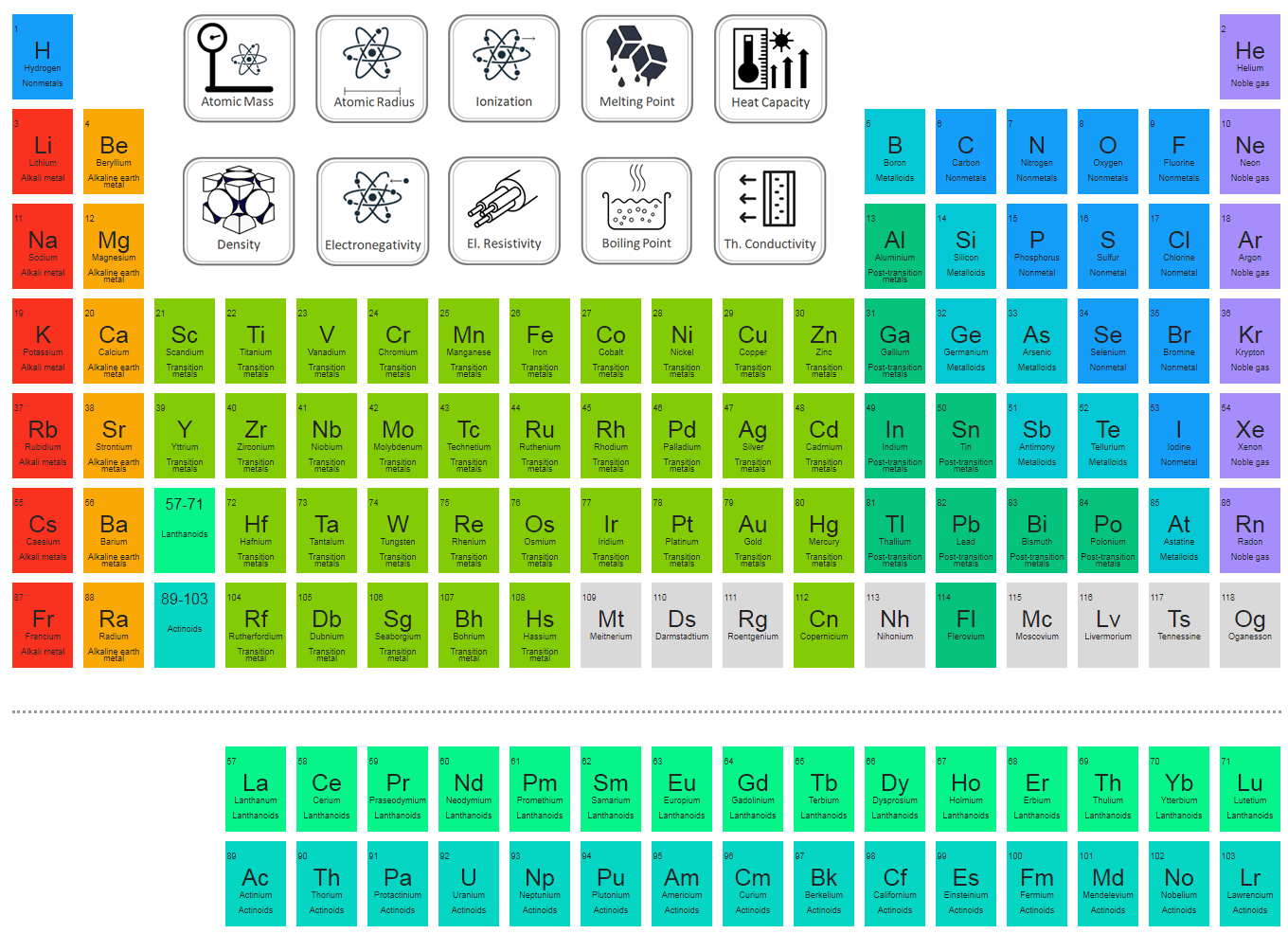
Both published tables with the elements arranged according to increasing atomic mass. Fluorine (F), chlorine (Cl), bromine (Br), and iodine (I) also exhibit similar properties to each other, but these properties are drastically different from those of any of the elements above.ĭimitri Mendeleev in Russia (1869) and Lothar Meyer in Germany (1870) independently recognized that there was a periodic relationship among the properties of the elements known at that time. For example: Li, Na, and K are much more reactive than are Ca, Sr, and Ba Li, Na, and K form compounds with oxygen in a ratio of two of their atoms to one oxygen atom, whereas Ca, Sr, and Ba form compounds with one of their atoms to one oxygen atom. However, the specific properties of these two groupings are notably different from each other. A second grouping includes calcium (Ca), strontium (Sr), and barium (Ba), which also are shiny, good conductors of heat and electricity, and have chemical properties in common. 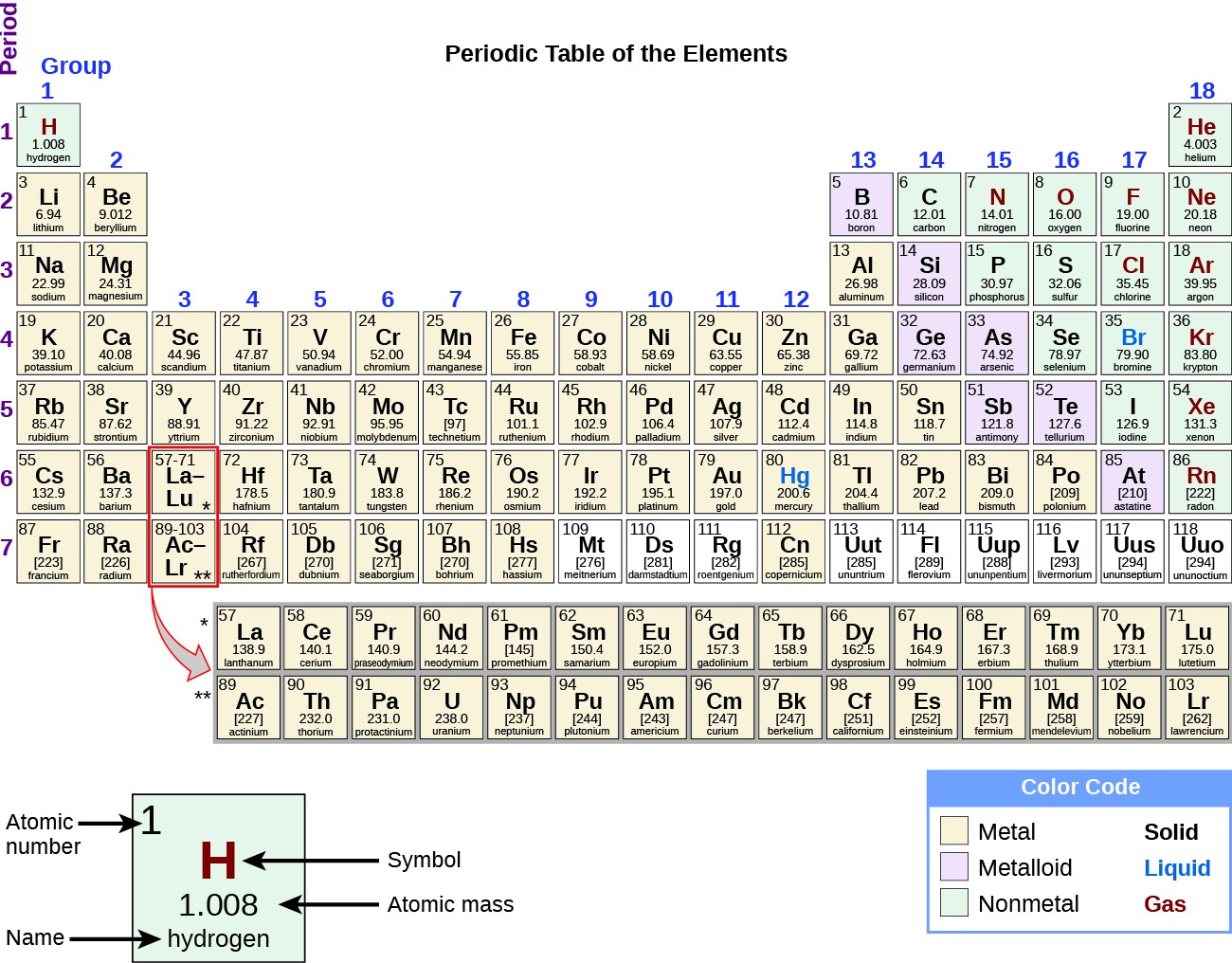
One such grouping includes lithium (Li), sodium (Na), and potassium (K): These elements all are shiny, conduct heat and electricity well, and have similar chemical properties. Identify metals, nonmetals, and metalloids by their properties and/or location on the periodic tableĪs early chemists worked to purify ores and discovered more elements, they realized that various elements could be grouped together by their similar chemical behaviors.Predict the general properties of elements based on their location within the periodic table.State the periodic law and explain the organization of elements in the periodic table.The third isotope, tritium, was discovered in 1934.Įstimated Crustal Abundance: 1.40×10 3 milligrams per kilogramĮstimated Oceanic Abundance: 1. The second, a stable isotope called deuterium, was discovered in 1932. The simplest isotope, called protium, is just ordinary hydrogen. Some of the common ones are: water (H 2O), ammonia (NH 3), methane (CH 4), table sugar (C 12H 22O 11), hydrogen peroxide (H 2O 2) and hydrochloric acid (HCl). Hydrogen combines with other elements to form numerous compounds. 
Liquid hydrogen is used in the study of superconductors and, when combined with liquid oxygen, makes an excellent rocket fuel. Hydrogen is also added to fats and oils, such as peanut oil, through a process called hydrogenation. Large amounts of hydrogen are combined with nitrogen from the air to produce ammonia (NH 3) through a process called the Haber process. Hydrogen is a commercially important element. The sun's supply of hydrogen is expected to last another 5 billion years. The same process, known as fusion, is being studied as a possible power source for use on earth. Hydrogen is the raw fuel that most stars 'burn' to produce energy. It is estimated that 90% of the visible universe is composed of hydrogen. Hydrogen was first recognized as a distinct element by Henry Cavendish in 1766.Ĭomposed of a single proton and a single electron, hydrogen is the simplest and most abundant element in the universe. Written records indicate that Robert Boyle produced hydrogen gas as early as 1671 while experimenting with iron and acids. Scientists had been producing hydrogen for years before it was recognized as an element. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
